Engineering Hormone Prosthetics for Autonomous Disease Management
Mission
Endocrine and metabolic diseases affect hundreds of millions of lives and demand constant monitoring, dosing, and decision-making.
My research focuses on developing autonomous hormone prosthetics. These are wearable and implantable devices that restore, modulate, or augment hormone signaling to reduce the burden of active disease management, expand treatment access, and improve quality of life.
Research
Engineering hormone prosthetics to improve access, adherence, and efficacy of chronic hormone therapies.
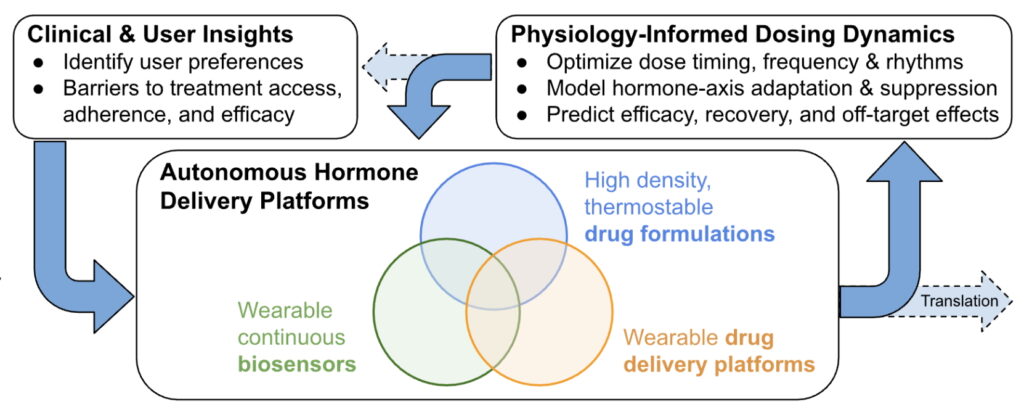
My approach integrates clinical and user insight, autonomous drug delivery technologies, and physiology-informed dosing strategies to reshape how chronic hormone therapies are designed and delivered.
Ultra-Long Acting Passive Delivery Devices
Biodegradable, ultra-long lasting drug delivery devices towards semi-autonomous hormone therapies.
Monolithic Shape-Shifting Absorbable Implants for Contraception (MoSAIC)
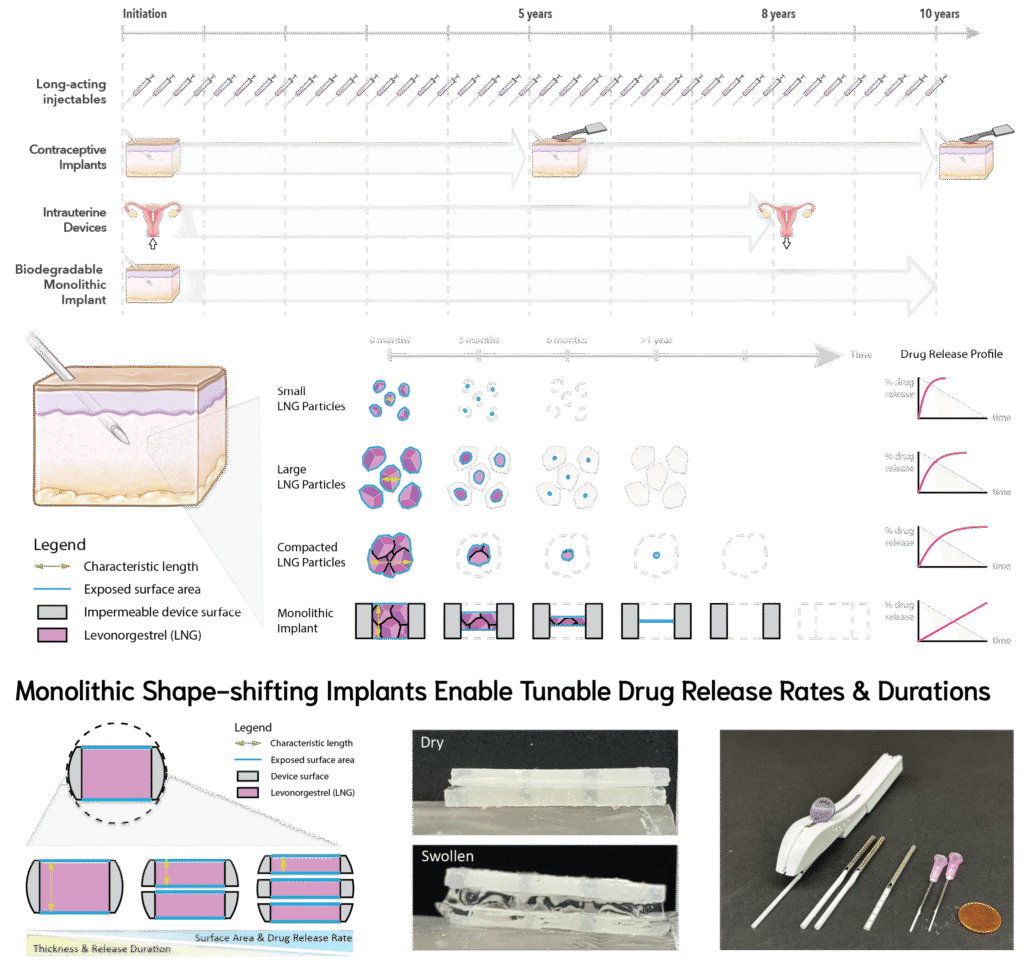
The Challenge: Long-acting contraceptive implants like Jadelle® and Nexplanon® are highly effective but require painful insertion and surgical removal, limiting access and adoption.
Our Solution: We developed MoSAIC, a contraceptive implant that is smaller, less invasive to insert, and fully bioresorbable, eliminating the need for surgical removal. MoSAIC uses two key innovations:
- Monolithic reservoir: space-efficient drug loading, zero-order release, and high efficient drug utilization.
- Shape-shifting design: expands after insertion to enable small-trocar delivery and independent control drug of release rate and duration.
Why it Matters: This architecture supports a range of hydrophobic drugs, making MoSAIC a generalizable platform for chronic drug therapy.
Related Publications: Link
Resorbable Sequential Timed Delivery (ReSTeD) Implants for GLP-1 Receptor Agonist Therapy
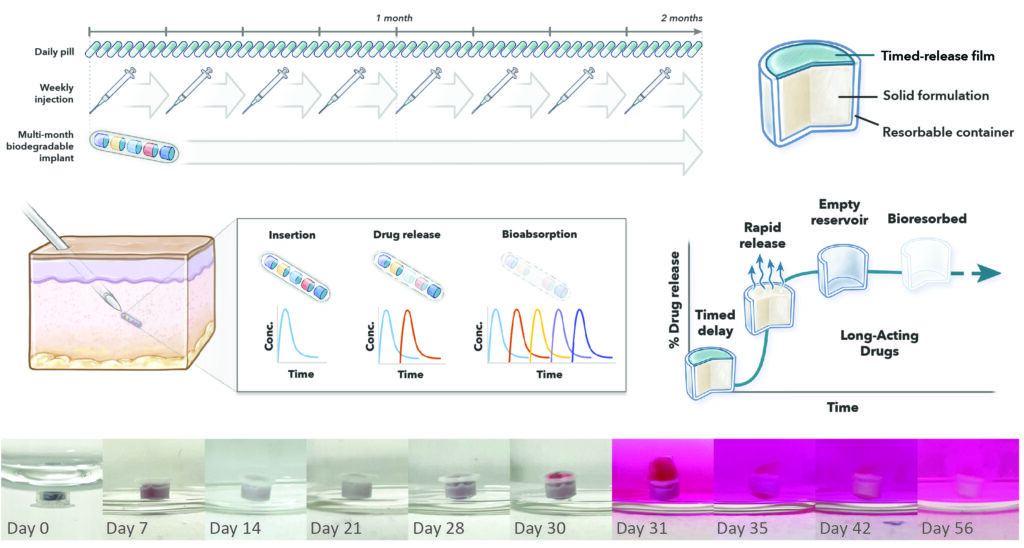
The Challenge: Long-acting GLP-1 receptor agonists such as semaglutide for diabetes and obesity remains hindered by once-weekly administration, contributing to poor medication adherence.
Our Solution: We developed ReSTeD implants, which autonomously deliver sequential doses of long-circulating semaglutide over multi-month timescales. ReSTeD employs:
- A space-efficient, multi-reservoir architecture in which encapsulated drug depots are programmed to release at weekly intervals, enabling automated, pulsatile dosing without patient involvement to mimic the pharmacokinetics of repeated dosing.
- Each reservoir contains a high-density compacted semaglutide formulation, while the device’s fully bioresorbable construction eliminates the need for surgical retrieval.
Why it Matters: ReSTED demonstrates a new approach for achieving multi-month delivery of peptide and protein therapeutics.
Continuous Biosensors & Active Delivery Devices
Real-time biochemical sensors & on-demand drug delivery actuators towards autonomous closed-loop therapies.
Hypoglycemia-Responsive Transdermal Microneedle Patches for Closed-Loop Glucagon Therapy
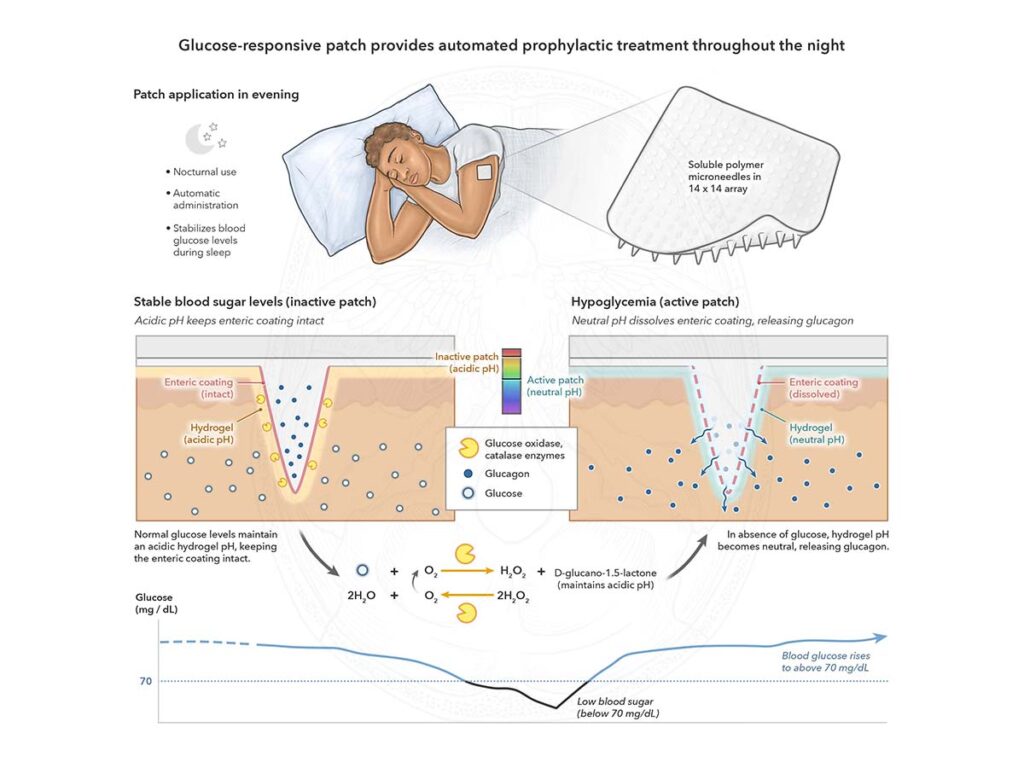
The Challenge: Hypoglycemia is a life-threatening complication of diabetes and a major barrier to effective insulin therapy. Although glucagon rapidly reverses hypoglycemia, current dosage forms require manual, painful and technically complex administration – making them undesirable and unusable for patients who are asleep, unconscious, or unaware of their condition.
Our Solution: We designed a wearable, glucose-responsive patch that provides automated glucagon release during nocturnal hypoglycemia. The responsive patch employs a glucose-oxidase-based enzymatic reaction that automatically triggers rapid glucagon release and continued elution under hypoglycemic conditions.
Why it Matters: We demonstrate a low-cost, autonomous delivery system that enables glucagon treatment in settings where intervention is currently impractical.
Related Publications: Link 1 | Link 2
Nano-FET Biosensor Architectures for High-Performance Biochemical Monitoring
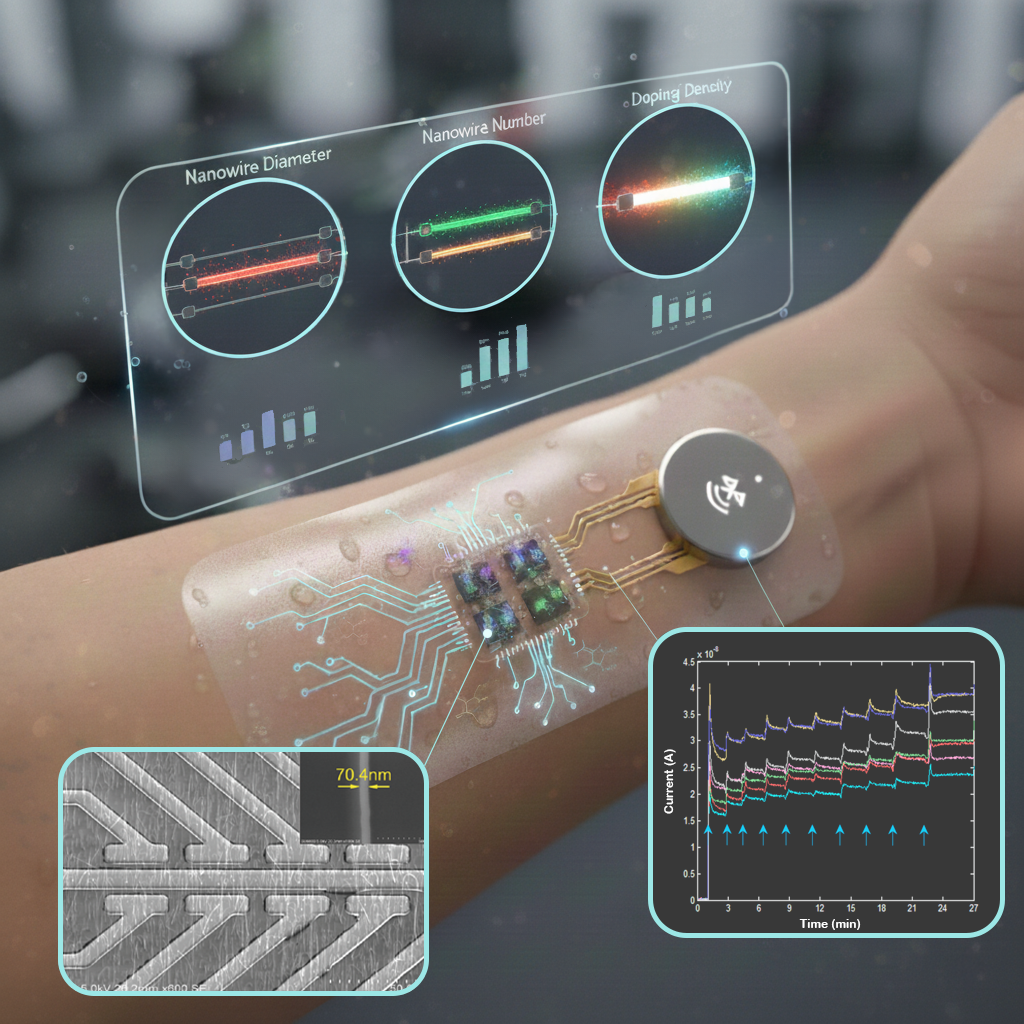
The Challenge: Nanowire field-effect transistor (nano-FET) biosensors enable ultra-sensitive, real-time, label-free biochemical measurement, making them a promising platform for continuous health monitoring. However, optimizing their performance requires new experimental methods to systematically study how device architecture shapes sensing behavior.
Our Solution: I developed a systematic experimental framework to map how nano-FET design parameters affect sensing performance. By testing how nanowire size, number, and electronic properties influence sensitivity, I show that thinner, lightly doped nanowires yield higher signal response, while multi-nanowire configurations introduce trade-offs between signal amplitude and noise.
Why it Matters: These insights establish a physics-based foundation for engineering reliable, high-performance nano-FET biosensors for real-world use.
Advanced Therapeutic Formulations
Next-generation formulations for improved treatment of chronic diseases.
Readily Soluble and Thermostable (ReST) Glucagon Formulations
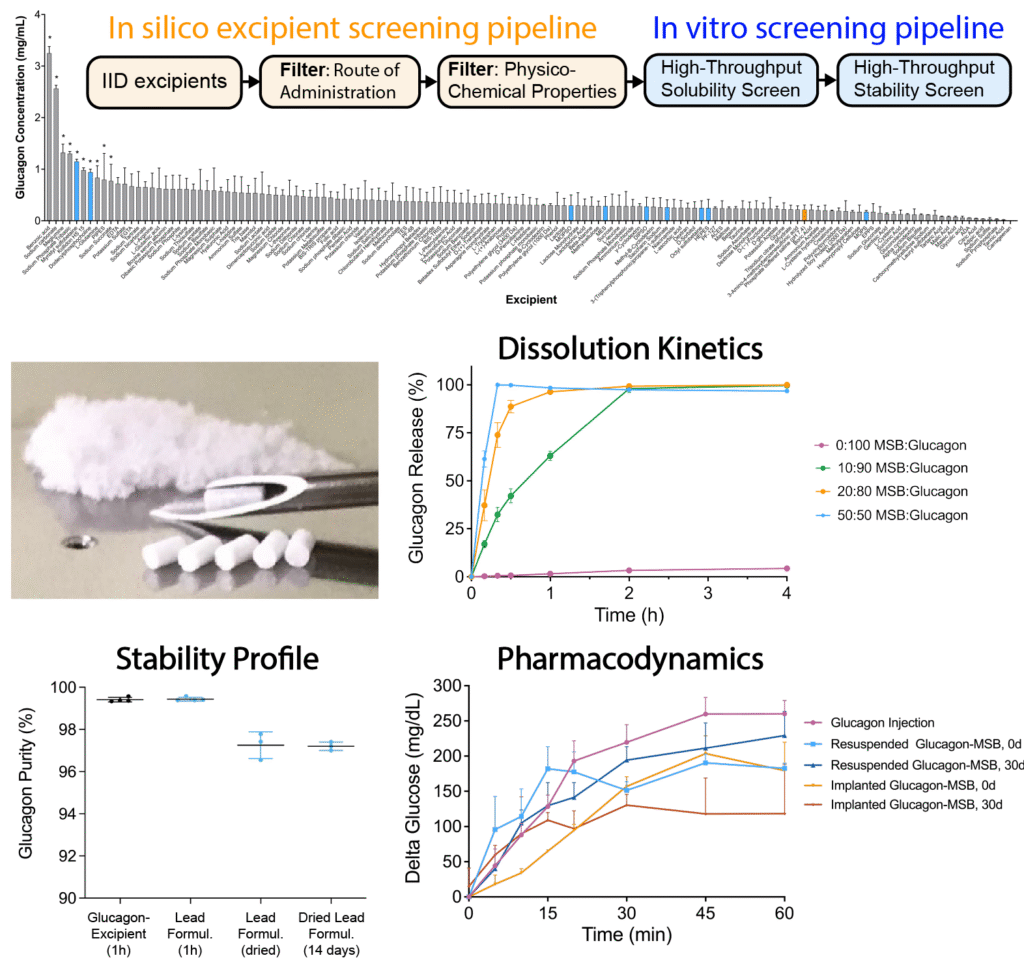
The Challenge: Developing next-generation therapies for hypoglycemia is hindered by the poor stability and solubility of glucagon. Its instability in aqueous solutions prevents long-term storage and use in infusion pumps, while its poor solubility in physiologic fluids prevents direct administration as a solid.
Our Solution: I developed solid Readily Soluble and Thermostable (ReST) glucagon formulations that are intrinsically stable yet rapidly dissolve in physiological fluids, enabling direct administration without reconstitution. Using a data-driven, high-throughput formulation approach, I identified drug-excipient combinations with rapid dissolution kinetics and high drug loading efficiency.
Why it Matters: ReST formulations enable development of compact, solid-state microneedle devices for painless, user-friendly glucagon delivery in the treatment of hypoglycemia.
Related Publications: Link
Thermostable Insulin Formulations for Long-Term Basal Insulin Therapy
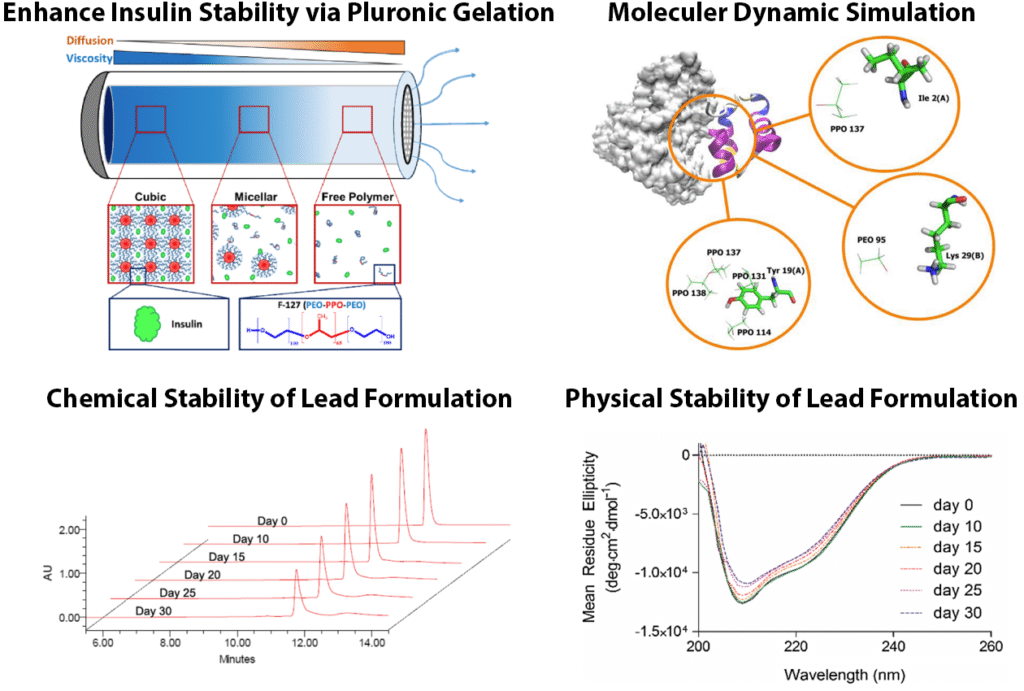
The Challenge: Wearable, ingestible and implantable drug-delivery devices offer promising options for long-term management of chronic diseases, however their treatment durations are limited by low drug loading capacities and the poor thermal stability of many peptide and protein drugs.
Our Solution: To address this limitations, I investigated the use of the non-ionic amphiphilic surfactant Pluronic F-127 for developing highly concentrated, thermostable formulations of insulin. Increasing the polymer concentration improved both the chemical and physical stability of insulin, enabling the formulation to remain stable for up to 30 days under simulated in vivo conditions. This enhanced stability arises from the copolymer’s amphiphilic interactions with insulin and the increased viscosity associated with gel formation.
Why it Matters: These findings provide a framework for creating high-density, thermostable peptide formulations suitable for long-acting drug delivery devices.
Related Publications: Link
Polymer-Lipid Nanoparticles for Targeted Delivery to Brain Tumors
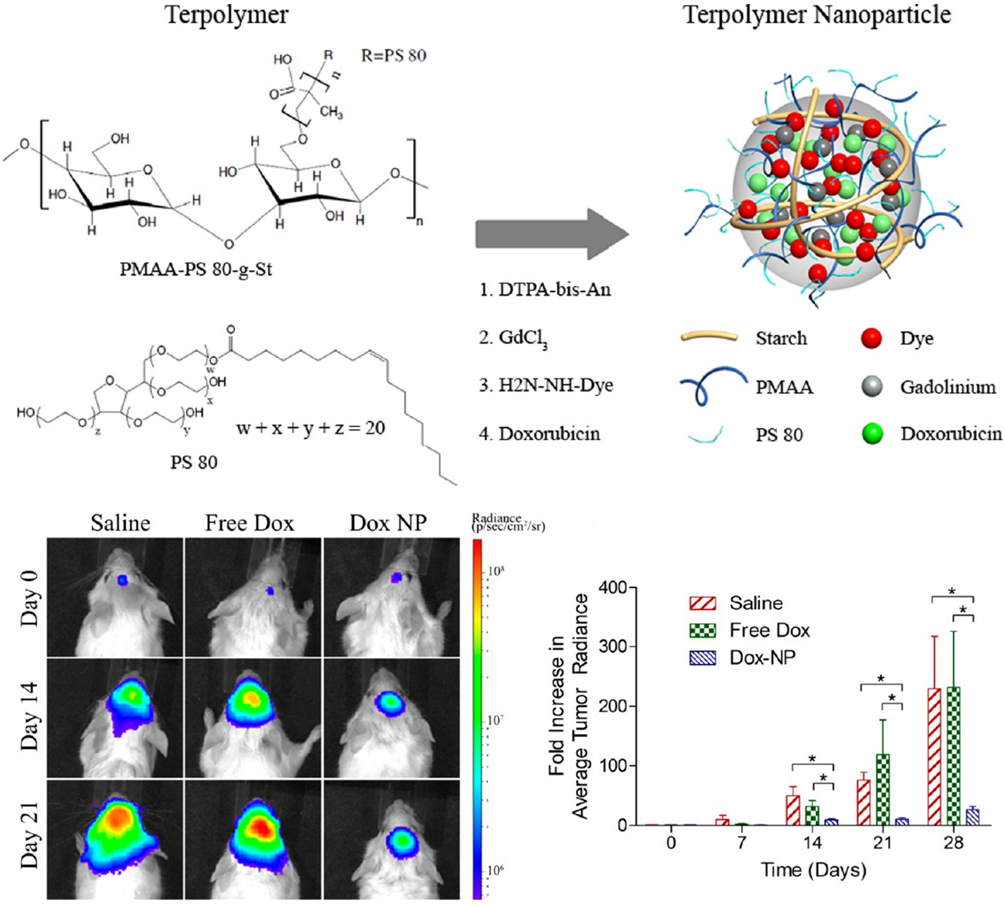
The Challenge: Chemotherapy treatments for brain cancers are often ineffective because most drugs cannot cross the blood–brain barrier (BBB) to reach tumor lesions at therapeutic levels.
Our Solution: I developed several nanoparticle-based delivery systems engineered to cross the BBB safely and efficiently. I evaluated these platforms using multiple anticancer agents commonly employed in oncology, including doxorubicin, docetaxel, and trastuzumab. In preclinical brain tumor models, these nanoparticles consistently achieved higher intratumoral drug concentrations and more reliable delivery to brain lesions than the drugs alone. T
Why it Matters: These findings highlight a promising strategy to enhance the effectiveness of systemic therapies for both primary and metastatic brain tumors and provide key design principles for engineering nanocarriers that achieve efficient and selective brain targeting.